international conference in endourology 2025, Rome, İtalya, 27 - 29 Kasım 2025, (Yayınlanmadı)
Cystinuria is a rare inherited disorder leading to recurrent cystine stone formation and significant morbidity, particularly in pediatric patients. Tiopronin is one of the mainstays of pharmacological therapy. Despite its widespread use, there is limited real-world safety data regarding tiopronin. This novel study aims to evaluate the adverse event profile of tiopronin using the FAERS database.
FAERS reports from 2014 to 2025 were analyzed. After deduplication according to FDA guidance, cases were included only if tiopronin was identified as the primary suspect drug. Adverse events were coded using MedDRA version 28.0 at both the Preferred Term (PT) and System Organ Class(SOC) levels. Four disproportionality algorithms—Reporting Odds Ratio, Proportional Reporting Ratio, Bayesian Confidence Propagation Neural Network, and Multi-item Gamma Poisson Shrinker—were used for signal detection. Events directly related to tiopronin’s indication were excluded to reduce confounding.
A total of 1,838 unique tiopronin-associated adverse event reports were identified. Pediatric patients (<18 years) accounted for 69.6% of cases, and most reports originated from the United States (99.4%). Reports were predominantly submitted by physicians (86.5%). Disproportionality analysis revealed significant safety signals in 26 SOCs and 79 PTs. In addition to expected events related to cystinuria and stone management, notable findings included significant associations with hyposmia, skin atrophy, breath odour, tongue discolouration, and incorrect dosage administered—none of which are currently included in the tiopronin product label. (Figure 1) The signal for incorrect dosage administered was particularly strong, highlighting challenges with long-term therapy and dosing in pediatric populations. AE reporting peaked in 2017, followed by a decline, likely reflecting changes in drug availability and utilization.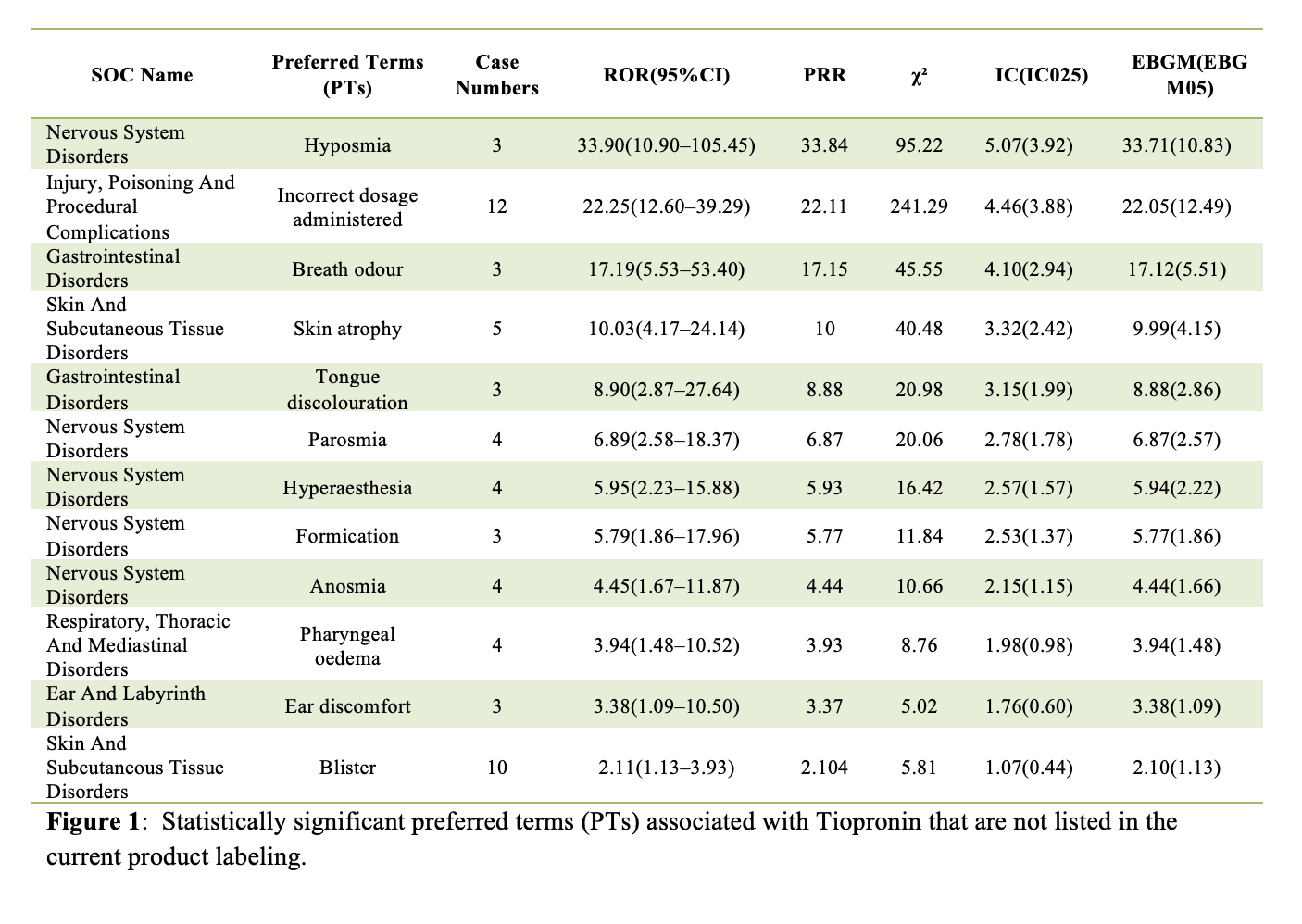
The analysis identified both expected and previously unrecognized safety signals, including several adverse events not captured in current product information.The predominance of pediatric cases and the high frequency of serious outcomes emphasize the need for enhanced pharmacovigilance, careful monitoring, and improved education for families and clinicians managing tiopronin therapy in children.